
- #GAMP 4 AND GAMP 5 GUIDELINES. ZIP FILE#
- #GAMP 4 AND GAMP 5 GUIDELINES. PATCH#
- #GAMP 4 AND GAMP 5 GUIDELINES. SOFTWARE#
- #GAMP 4 AND GAMP 5 GUIDELINES. ZIP#
- #GAMP 4 AND GAMP 5 GUIDELINES. DOWNLOAD#
The first version was written by a group of volunteers in the UK in the early 1990s as a mechanism to control suppliers of process equipment to the pharmaceutical industry, and this legacy survived through to version 4. The problem lay in the origins of the GAMP guide. For example, when a commercially available laboratory system was validated, functional and design specifications were written for virtually no gain but at a great cost in time and resources.
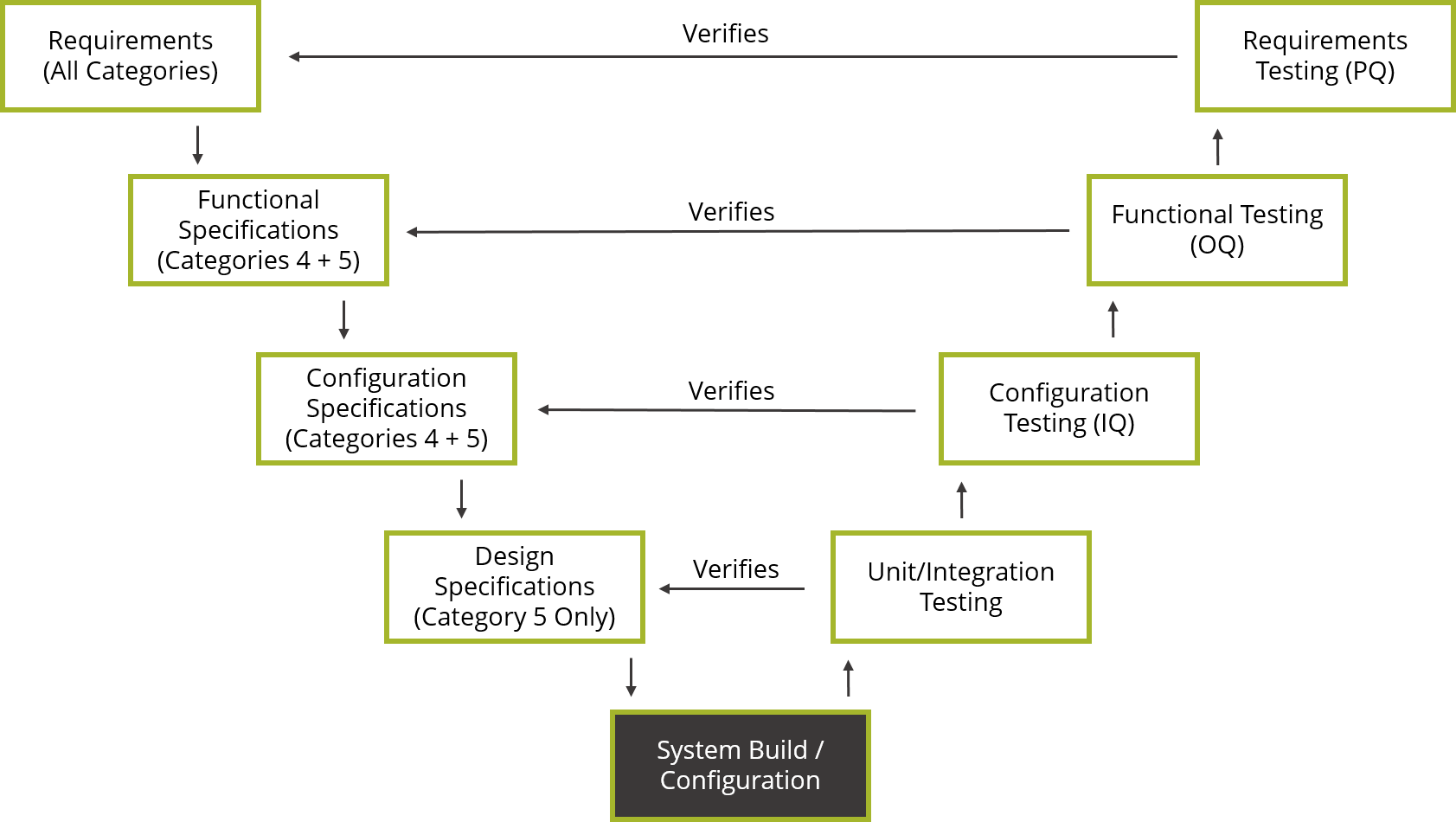
Therefore, every validation was shoehorned into an inappropriate model because there was little thought and intelligence applied and the model followed blindly. It had very little to do with computerized systems, especially laboratory ones. My rationale for this position is that versions 1–4 of the guide presented a single life cycle V model that was really only applicable to process equipment and manufacturing systems.
#GAMP 4 AND GAMP 5 GUIDELINES. SOFTWARE#
I love the classification of software outlined in Appendix M4 and hate the life cycle V model. However, before we continue much further I should also declare a vested interest: I have a love–hate relationship with the GAMP guide. We will explore version 5 of the software classification and see what changes we need to make to ensure that it can be implemented practically and effectively in any laboratory. This is one of the best parts of the guide as it has an in-built risk assessment, as we shall see in this column. This publication has been available since 1994, when version 1 was informally published in the UK, and since its inception it has always contained a classification of software. Version 5 of the Good Automated Manufacturing Practice (GAMP) guide (1) was released last year. This column will look at what the changes mean for the laboratory and whether all of these should be implemented.
#GAMP 4 AND GAMP 5 GUIDELINES. ZIP#
A detailed list of contents is included in the downloadable Zip file.ġ.6.1 Overview of GAMP Documentation StructureĢ.1.4 Science Based Quality Risk Managementģ.3 Computerized System Validation FrameworkĤ.2.2 Specification, Configuration, and CodingĤ.3.2 Service Management and Performance Monitoringĥ.2 Science Based Quality Risk ManagementĦ.1.1 Computerized Systems Policies and ProceduresĦ.1.2 Identifying Clear Roles and ResponsibilitiesĦ.2.5 Determine Strategy for Achieving Compliance and Fitness for Intended UseĦ.2.8 Development and Review of Software for Custom ApplicationsĦ.2.11 Maintaining System Compliance During Operationħ.1 Supplier Products, Applications, and Servicesħ.1.1 Non-Configured Product (GAMP Category 3)ħ.1.2 Configured Product (GAMP Category 4)ħ.1.3 Custom Application (GAMP Category 5)ħ.13 System Support and Maintenance During OperationĨ.1 Establishing Verifiable and Objective User RequirementsĨ.8 Anticipating Data Archiving and Migration Needsġ0 International Standards and Certificationģ Contents of the Quality and Project Plan
#GAMP 4 AND GAMP 5 GUIDELINES. ZIP FILE#
A Zip file with these supporting materials will be provided to purchasers through the ISPE Bookstore.
#GAMP 4 AND GAMP 5 GUIDELINES. DOWNLOAD#
Valuable supporting materials, including key diagrams, templates, forms, example documents, and background information is available to download for purchasers of GAMP 5.
#GAMP 4 AND GAMP 5 GUIDELINES. PATCH#
It contains new information on outsourcing, electronic batch recording, end user applications (such as spreadsheets and small database applications), and patch management. This revolutionary Guide addresses the entire lifecycle of an automated system and its applicability to a wide range of information systems, lab equipment, integrated manufacturing systems, and IT infrastructures. It points to the future of computer systems compliance by centering on principles behind major industry developments such as PQLI ICH Q8, Q9, Q10 and ASTM E2500. This technical document describes a flexible risk-based approach to compliant GxP regulated computerized systems, based on scalable specification and verification. GAMP ® 5 provides pragmatic and practical industry guidance to achieve compliant computerized systems fit for intended use in an efficient and effective manner.

Author(s): Bredesen, Anders Brooks, Sam Buffi, Jay Cappucci, Winnie Cherry, Mark Clark, Chris Evans, Gail Hambloch, Heinrich Jones, Colin Kane, Paige Margetts, Tony Perez, Arthur "Randy" Robertson, Peter Samways, Kate Selby, David Wingate, Guy Wyn, Sion
